http://www.lookingforalternatives.com.au/chemical-soft-drinks/
this articles really show the detailed facts of the nutritional value of soft drink. it contain so many different chemical that individually can cause damage. even though certain soft drink is good for you, it is only under special circumstances and only in low dosage. therefore we felt that we should cut down our amount of soft drink and should only be drank occasionally but never a replacement of water.
Monday, 30 July 2012
Caffeine
caffeine acts as a nervous system stimulant, temporarily warding off drowsiness and restoring alertness. at low dosage, it can actually help the human body in many ways. however, it can be toxic at high dosage and iot can lead to cancer or even death.
here is some of the soft drink that contain caffieine and its amount:
fanta-0mg of caffeine in a can
coca cola-34mg of caffeine in a can
Diet coke-45mg of caffeine in a can
McDonald's Coffee (large)-145mg in a 16oz cups
Mountain Dew-54mg of caffeine in a can
Pepsi-69mg of caffeine in a can
Red Bull-120mg of caffeine in a can
Starbucks Grande Coffee-330mg of caffeine in a 16oz cups
amount of caffeine needed for people vary for each person. this is a website to find out how much caffeine you can take.
http://www.energyfiend.com/death-by-caffeine
generally, the amount of caffeine to considered overdose is around 300-500mg
here is some of the soft drink that contain caffieine and its amount:
fanta-0mg of caffeine in a can
coca cola-34mg of caffeine in a can
Diet coke-45mg of caffeine in a can
McDonald's Coffee (large)-145mg in a 16oz cups
Mountain Dew-54mg of caffeine in a can
Pepsi-69mg of caffeine in a can
Red Bull-120mg of caffeine in a can
Starbucks Grande Coffee-330mg of caffeine in a 16oz cups
amount of caffeine needed for people vary for each person. this is a website to find out how much caffeine you can take.
http://www.energyfiend.com/death-by-caffeine
generally, the amount of caffeine to considered overdose is around 300-500mg
HPLC Results of Soft Drink Additives
The attached chromatogram shows multiple soft drinks and a standard, showing which components are present in each soft drink.
(Source: http://www.dionex.com/en-us/images/page-cgram/img-82605-21531.jpg)For (A) Minute Maid Orange Soda, peaks 2, 7, and 8 match, indicating the presence of citric acid, sorbate potassium, and benzoic acid.
For (B) Diet Coke, peaks 5, 6, and 8, match, indicating the presence of Caffeine, Aspartame (an artificial sweetener), and benzoic acid
For (C) Mountain Dew, peaks, 2, 5, and 8 match, indicating the presence of Citric acid, Caffeine, and Benzoic Acid.
The chromatogram attached below shows a HPLC of the sweeteners present in a soft drink.
(Source: http://www.shimadzu.com/an/industry/foodbeverages/qn50420000001y0d-img/qn504200000029me.gif)The chromatogram attached below shows a HPLC of canned coffee, canned tea, and canned orange juice, along with the monosaccharides present in each of them.
(Source: http://www.hplc.com/shodex/img/s0928.gif)
Sodium Benzoate/Benzoic Acid
Sodium benzoate is a common food preservative. In soft drinks, it is dissolved to form benzoic acid. It is also the main cause for benzene content in soft drinks. As benzene is a carcinogen, ingesting soft drinks preserved with sodium benzoate may increase the risk of cancer.
However, benzene is only released from sodium benzoate under certain environmental conditions, therefore foods preserved with sodium benzoate are usually stored away from light in a cool dark environment.
However, benzene is only released from sodium benzoate under certain environmental conditions, therefore foods preserved with sodium benzoate are usually stored away from light in a cool dark environment.
Sunday, 29 July 2012
HPLC-MS
In HPLC-MS (High Performance Liquid Chromatography), the components in the analyte are analyzed through the use of a Mass Spectrometer detector.
A Mass Spectrometer is a detector that measures the mass-to-charge ratio of the eluting components. This provides a very accurate identification of the components due to the high improbability of two different compounds of similar retention times having similar mass-to-charge ratios.
A drawback of the Mass Spectrometer detector is that it is a destructive detector, therefore the samples cannot be recovered after analysis.
A Mass Spectrometer is a detector that measures the mass-to-charge ratio of the eluting components. This provides a very accurate identification of the components due to the high improbability of two different compounds of similar retention times having similar mass-to-charge ratios.
A drawback of the Mass Spectrometer detector is that it is a destructive detector, therefore the samples cannot be recovered after analysis.
HPLC-PDA
In HPLC-PDA, components are not only separated through their retention times (affinity to the stationary phase), but they are further analyzed through the use of a Photo Diode Array detector.
How does a Photo Diode Array detector work?
A spectrum of light, usually within the UV-vis region is shone onto the sample and the absorbance of the sample to each wavelength within the spectrum is measured simultaneously to determine the absorbance of the eluting components instantly at any point of time during the analysis.
Compared to UV-vis spectrophotometry, this is much more efficient for analyzing components within the same mixture which have different lambda-max wavelengths as a spectrum is shone onto the sample as compared to a monochromatic beam of light in UV-vis spectrophotometry.
This detector is extremely useful for determining the identity of each component, as it is highly unlikely for different compounds which have similar retention times, to absorb within the same wavelengths as well. This is also a non-destructive detector, therefore the samples may be reused if needed.
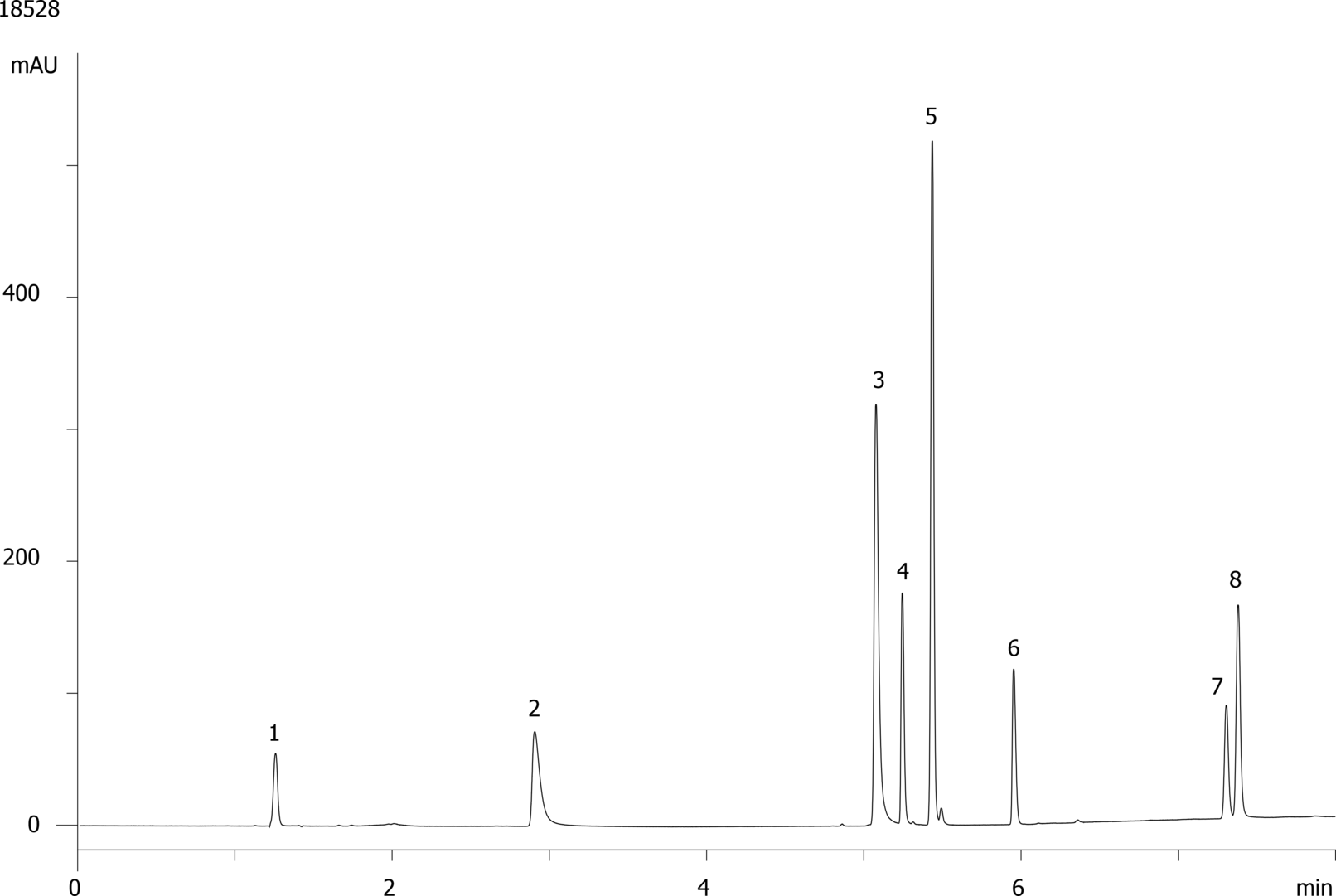
A chromatogram of a HPLC-PDA of a soft drink is attached below:
(Source: 18528.gif)
Peak 1: Ascorbic Acid
Peak 2: Acesulfame K
Peak 3: Saccharin
Peak 4: Quinine
Peak 5: Caffeine
Peak 6: Aspartame
Peak 7: Sorbic Acid
Peak 8: Benzoic Acid
How does a Photo Diode Array detector work?
A spectrum of light, usually within the UV-vis region is shone onto the sample and the absorbance of the sample to each wavelength within the spectrum is measured simultaneously to determine the absorbance of the eluting components instantly at any point of time during the analysis.
Compared to UV-vis spectrophotometry, this is much more efficient for analyzing components within the same mixture which have different lambda-max wavelengths as a spectrum is shone onto the sample as compared to a monochromatic beam of light in UV-vis spectrophotometry.
This detector is extremely useful for determining the identity of each component, as it is highly unlikely for different compounds which have similar retention times, to absorb within the same wavelengths as well. This is also a non-destructive detector, therefore the samples may be reused if needed.
A chromatogram of a HPLC-PDA of a soft drink is attached below:
(Source: 18528.gif)
Peak 1: Ascorbic Acid
Peak 2: Acesulfame K
Peak 3: Saccharin
Peak 4: Quinine
Peak 5: Caffeine
Peak 6: Aspartame
Peak 7: Sorbic Acid
Peak 8: Benzoic Acid
HPLC: High-Performance Liquid Chromatography
High-performance liquid chromatography, also known as high-pressure liquid chromatography, is an analytical chromatography technique used to separate mixtures into their components through the various differences in affinity of the components to the stationary phase (physico-chemical properties). This causes a difference in retention times for the different components.
However, retention times alone are not enough to determine the identity of the components, a detector is required to conclude the identity of the components.
There are many detectors available for HPLC systems, although we will only be covering two detectors in the subsequent posts:
The Photo Diode Array detector (PDA)
The Mass Spectrometer detector (MS)
Analysis of Soft Drinks
There are many ways to analyze the composition of soft drinks.
Here are some examples:
HPLC-PDA
HPLC-MS
UV-vis Spectrophotometry (tentative, not recommended)
The methods will be discussed individually in the subsequent posts.
Here are some examples:
HPLC-PDA
HPLC-MS
UV-vis Spectrophotometry (tentative, not recommended)
The methods will be discussed individually in the subsequent posts.
Tartazine: What is it?
Tartrazine is a common food dye used in many soft drinks. It is usually used to produce a yellow coloring, although it is commonly mixed with other dyes to produce other colors.
Tartrazine is an artificial food coloring additive that is a cheap alternative to beta-cartotene, which is a natural and healthier food coloring additive.
What are the potential health risks posed by tartrazine intake?
Tartrazine is known to cause hyperactivity in children; increase the risk of Carpal Tunnel Syndrome; migraines; ADHD; and many other health problems.
Allergic reactions may also occur in people with high tartrazine sensitivity.
Some soft drinks that contain tartrazine:
Mountain Dew
Fanta
Tartrazine is an artificial food coloring additive that is a cheap alternative to beta-cartotene, which is a natural and healthier food coloring additive.
What are the potential health risks posed by tartrazine intake?
Tartrazine is known to cause hyperactivity in children; increase the risk of Carpal Tunnel Syndrome; migraines; ADHD; and many other health problems.
Allergic reactions may also occur in people with high tartrazine sensitivity.
Some soft drinks that contain tartrazine:
Mountain Dew
Fanta
Sunday, 15 July 2012
Exactly How Much Sugar Is Present In Our Soft Drinks?
http://www.sugarstacks.com/beverages.htm
The above website quantifies the mass of sugar used in various soft drinks in the form of sugar cubes, the images may help put into perspective the true amount of sugar in such soft drinks.
For example, a 355 ml can of Coca Cola contains 39 g of sugar, which is roughly equivalent to 9.5 sugar cubes, and a 1 liter bottle of the same soft drink contains 108 g of sugar, which is approximately 27.5 sugar cubes.
The above website quantifies the mass of sugar used in various soft drinks in the form of sugar cubes, the images may help put into perspective the true amount of sugar in such soft drinks.
For example, a 355 ml can of Coca Cola contains 39 g of sugar, which is roughly equivalent to 9.5 sugar cubes, and a 1 liter bottle of the same soft drink contains 108 g of sugar, which is approximately 27.5 sugar cubes.
(Source: http://www.sugarstacks.com/img/colas.jpg)
Friday, 13 July 2012
What Are Soft Drinks?
Soft drinks are beverages which are composed mainly of water, sweeteners and, flavoring agents of natural or artificial source. It also contains many common chemicals, including: sugars, carbon dioxide and caffeine.
However, there are still many unknown chemicals which are present in soft drinks.
What are they, and how can they affect our bodies?
However, there are still many unknown chemicals which are present in soft drinks.
What are they, and how can they affect our bodies?
Subscribe to:
Comments (Atom)