In HPLC-PDA, components are not only separated through their retention times (affinity to the stationary phase), but they are further analyzed through the use of a Photo Diode Array detector.
How does a Photo Diode Array detector work?
A spectrum of light, usually within the UV-vis region is shone onto the sample and the absorbance of the sample to each wavelength within the spectrum is measured simultaneously to determine the absorbance of the eluting components instantly at any point of time during the analysis.
Compared to UV-vis spectrophotometry, this is much more efficient for analyzing components within the same mixture which have different lambda-max wavelengths as a spectrum is shone onto the sample as compared to a monochromatic beam of light in UV-vis spectrophotometry.
This detector is extremely useful for determining the identity of each component, as it is highly unlikely for different compounds which have similar retention times, to absorb within the same wavelengths as well. This is also a non-destructive detector, therefore the samples may be reused if needed.
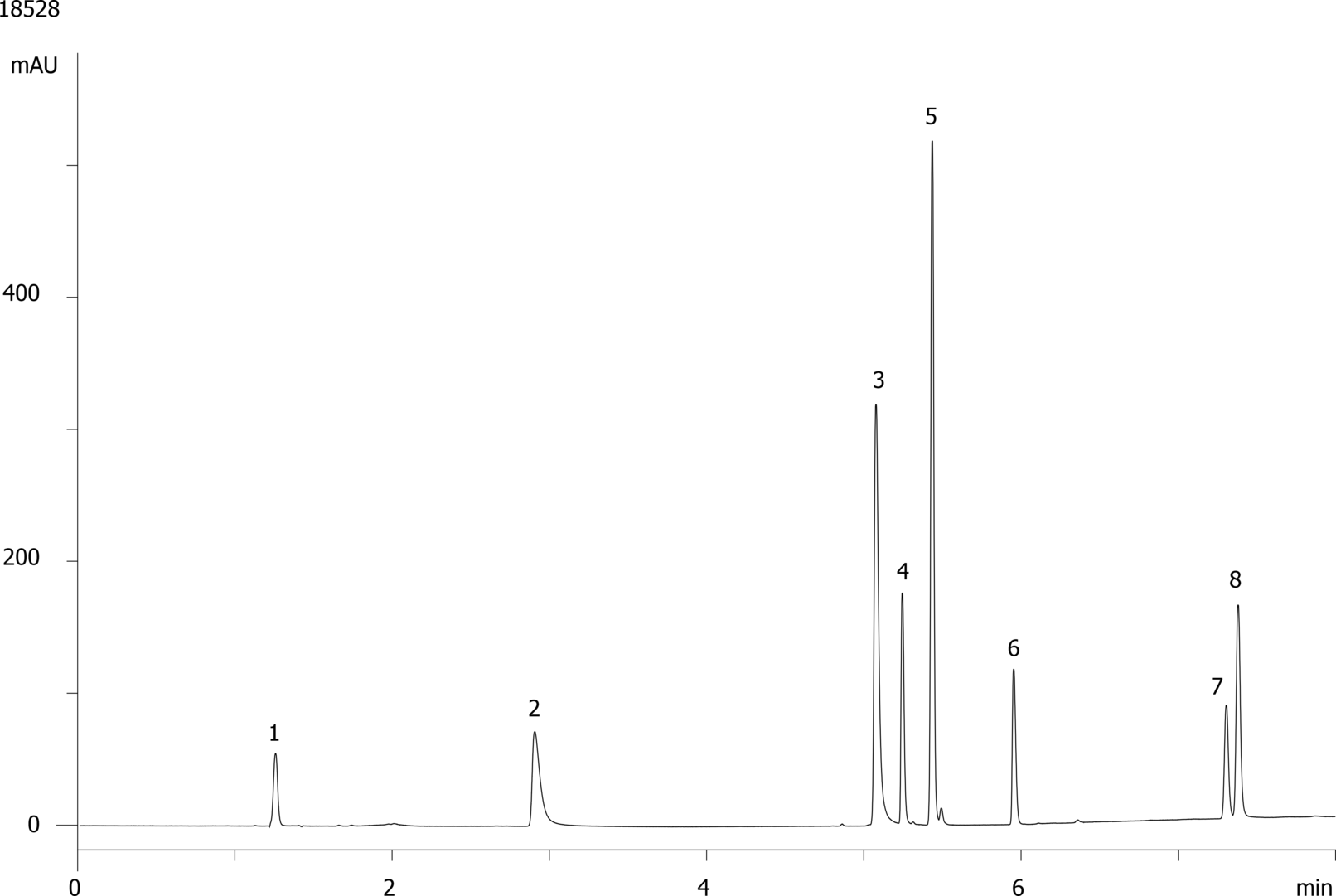
A chromatogram of a HPLC-PDA of a soft drink is attached below:
(Source: 18528.gif)
Peak 1: Ascorbic Acid
Peak 2: Acesulfame K
Peak 3: Saccharin
Peak 4: Quinine
Peak 5: Caffeine
Peak 6: Aspartame
Peak 7: Sorbic Acid
Peak 8: Benzoic Acid
you are clear my mind actually after reading your article i got clear my complete doubt. thanks for such easy understanding post. I also got some similar at here just for your info i post here link may be useful for future aspect how do photodiode arrays work?
ReplyDelete